$7.9 Million in NIH Awards Propel UMD Aging Research
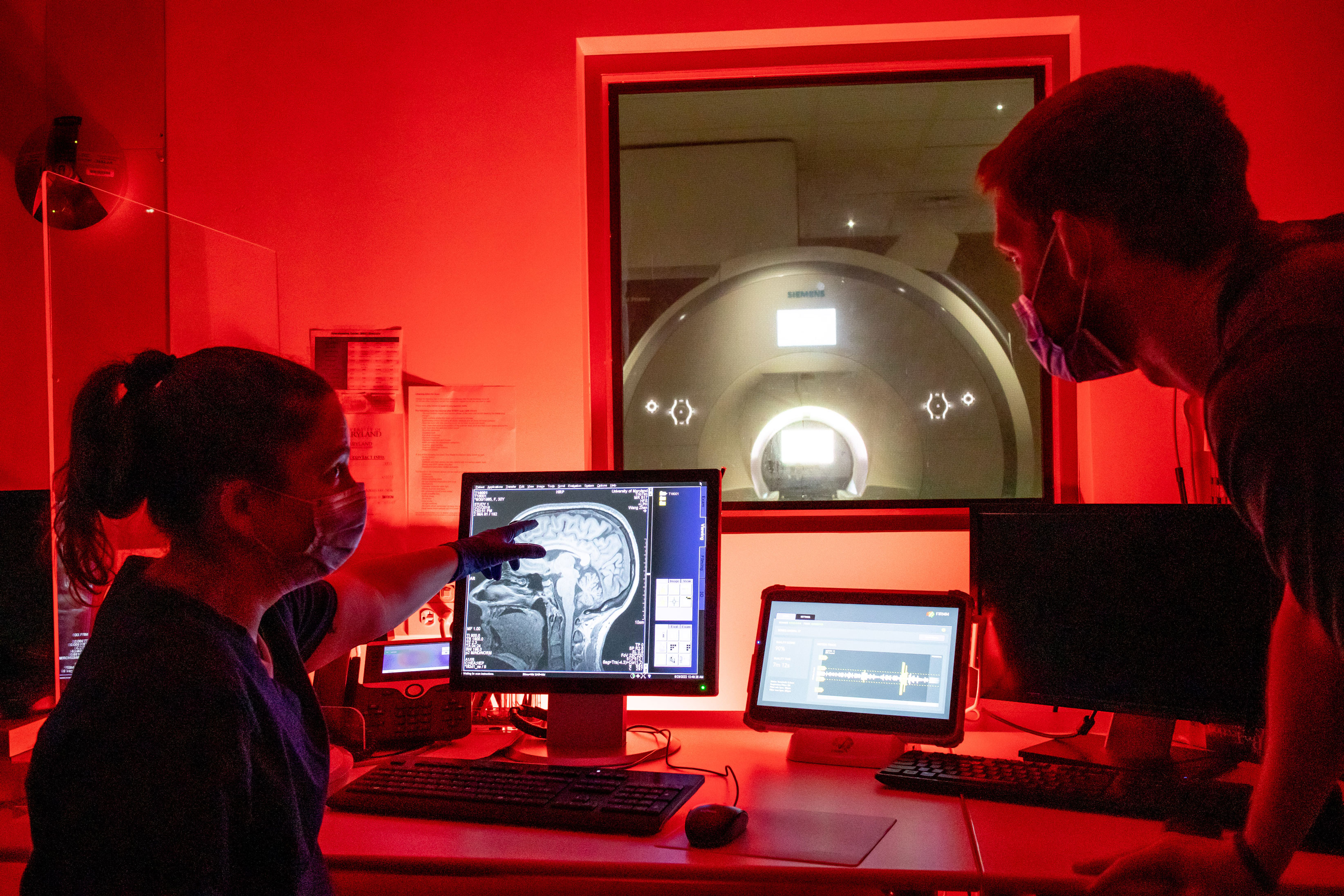
Aging is inevitable. But the development of Alzheimer’s disease (AD) and age-related dementias is not. This belief drives Brain and Behavior Institute (BBI) researchers at UMD to continue to innovate solutions to some of the toughest challenges related to Alzheimer’s and aging. These new basic science approaches, drawing from the life sciences, computer science and engineering, seek to translate to better prevention, diagnosis and treatment for problems in the human brain. An estimated 6.2 million Americans suffer from AD, with the number of deaths caused by Alzheimer’s more than doubling since 2000. For patients and loved ones, the disease exacts a staggering emotional and economic toll. “My grandmother was diagnosed with Alzheimer’s at age 59 and lived until the age of 73,” said Daniel Callow, a Ph.D. student in the Program in Neuroscience and Cognitive Science whose research seeks to understand the structural and functional networks of the aging brain, “So I know the degree of social isolation, economic cost and emotional turmoil that individuals go through when caring for someone with AD. It can be truly life-altering.” Callow’s research on the microstructure of the hippocampus in the aging brain is advised by J. Carson Smith, a professor in the Department of Kinesiology, who studies whether lifestyle factors such as exercise can be protective against Alzheimer’s for individuals with a genetic predisposition to the disease. An individual is at greater risk for developing AD depending on what version of the apolipoprotein E gene (APOE) they have inherited. The 25% of the population that carry the APOE4 variant are particularly vulnerable to developing Alzheimer’s. Funded by a five-year, $3.6 million award from the National Institute on Aging (NIA), Smith examines how exercise may delay or prevent the onset of Alzheimer’s and how exercise interventions can be effective against presymptomatic biomarkers of AD. Using fMRI scanning on healthy older adults with APOE4, Smith investigates whether exercise can improve performance on cognitive and neuroimaging tasks that are generally predictive of AD. Smith’s collaborators include James Barrett, a research associate of anesthesiology at the University of Maryland, Baltimore (UMB), and UMD engineering faculty Alisa Clyne, a professor in the Fischell Department of Bioengineering, and Ganesh Sriram, an associate professor in the Department of Chemical and Biomolecular Engineering. Smith, Clyne and Sriram first began their collaboration in 2020 on work that was funded by a BBI seed grant. It’s this connection to human patients, Clyne said, that excites her bioengineering research. “How can we help people from all backgrounds better understand how to live healthy lives so that we have an equitable society in terms of our physical and mental health?” asked Clyne. “The idea that you’re going to take a pill and never have a heart attack or suffer from Alzheimer’s disease is just not practical. The solutions for these problems will be about lifestyle.” Until recently, Clyne’s vascular mechanics laboratory focused primarily on interactions among cells and proteins of the cardiovascular system. But when some of her graduate students expressed interest in working on the brain, Clyne was enthused: the risk factors for cardiovascular disease—high cholesterol, high blood pressure, diabetes—are also known risk factors for Alzheimer’s disease. Clyne believes that there is a strong vascular component to AD progression. While it is known that the brains of people with AD show reduced glucose uptake, Clyne’s research focuses on the cells in charge of shuttling glucose into the brain. The Clyne lab studies this problem by engineering brain endothelial cells and comparing how metabolism differs in those with the APOE4 allele. “What motivates me is the idea that we could give people personalized recommendations for how to maintain their cardiovascular and neurological health for as long as possible,” said Clyne. Clyne and Sriram also collaborate with Thomas Longden, an assistant professor in the Department of Physiology at UMB, to study brain hypoperfusion and how the mechanics of low blood flow impact human performance. BBI researchers are also innovating diagnostic tools for Alzheimer’s. Ricardo Araneda a professor in the Department of Biology, looks to establish the loss of sense of smell as a biomarker for AD, since patients that develop dementia often lose their sense of smell before the onset of memory loss. This research is part of the Araneda laboratory’s investigation of neuromodulatory transmitters—that is, substances in the brain such as noradrenaline and dopamine that can tune the activity of diverse neural circuits. Basic research has proven that dysregulation of neuromodulatory transmitters underlies a range of neuropsychiatric disorders, including depression and anxiety. In order to establish diminished olfaction as a biomarker for AD, Araneda must first distinguish AD-related loss of smell from olfactory decline that is a product of normal healthy aging. This is the focus of Araneda’s five-year, $1.55 million award from the NIA, which supports a collaboration with UMB researchers Joseph Kao, a professor of in the Department of Physiology, and Adam Puche, a professor in the Department of Anatomy and Neurobiology. Intriguingly, their work is also relevant in the context of the long-term effects of COVID-19. “Because COVID-19, like Alzheimer’s disease, is also associated with olfactory dysfunction, there is some evidence that the SARS-CoV-2 virus may affect the same olfactory areas that are affected in AD," noted Araneda. The impact of Alzheimer’s disease is so widespread that it can be difficult for scientists to define the best research approaches. Enter Michael Cummings a professor in the Department of Biology and director of the Center for Bioinformatics and Computational Biology, whose applications of machine learning to biological research helps scientists define their domains of inquiry. Cummings’ research in machine learning is grounded in hypothesis generation. Whereas hypothesis testing is confirmatory, beginning with an informed supposition and seeking to determine whether or not this is true, hypothesis generation starts with a set of data and looks for patterns and associations. Cummings’ machine learning analyses help to challenge the limits of biological inferences and rejigger entry points for inquiry. In these endeavors, Cummings noted, collaboration is vital. “I’m not a neurobiologist, and I’m not an Alzheimer’s researcher. I’m a scientist who does bioinformatics and computational biology, among other things, so I always work with a team when developing machine learning approaches to analyze data.” Recently, Cummings’ collaboration with UMB investigators Maureen Kane, a professor in the Department of Pharmaceutical Science, and Marta Lipinski, an associate professor in the Department of Anesthesiology, yielded a four-year $2.7 million award from the NIA. He is also the recipient of a 2022 BBI seed grant award for his collaboration with Matthew Goupell, a professor in the Department of Hearing and Speech Sciences at UMD. The best collaborations extend Cummings’ capacities as much as he assists his colleagues in hypothesis generation. “Early on in my career, I would develop techniques and just turn the crank, so to speak, but that’s not the case for me now,” said Cummings. “Now my approach to challenging problems is more like art. In sculpture or painting, the artist conceives of something at some level of detail—even if crude or abstract—and then starts working with and growing through the medium. The best collaborations are like that. They push me and make me stretch.” “I ponder these problems when I walk. I think about them when I commute to campus. Those are the ideal collaborations.” ### Writer: Nathaniel Underland, underlan@umd.edu About the Brain and Behavior Institute: The mission of the BBI is to maximize existing strengths in neuroscience research, education and training at the University of Maryland and to elevate campus neuroscience through innovative, multidisciplinary approaches that expand our research portfolio, develop novel tools and approaches and advance the translation of basic science. A centralized community of neuroscientists, engineers, computer scientists, mathematicians, physical scientists, cognitive scientists and humanities scholars, the BBI looks to solve some of the most pressing problems related to nervous system function and disease.
Related Articles: September 9, 2022 Prev Next |